Pd-catalyzed asymmetric oxidative C-H/C-H cross-coupling reaction between dialkylaminomethylferrocenes and indolizines - ScienceDirect

Kinetic Resolution of Allyltriflamides through a Pd-Catalyzed C–H Functionalization with Allenes: Asymmetric Assembly of Tetrahydropyridines | Journal of the American Chemical Society

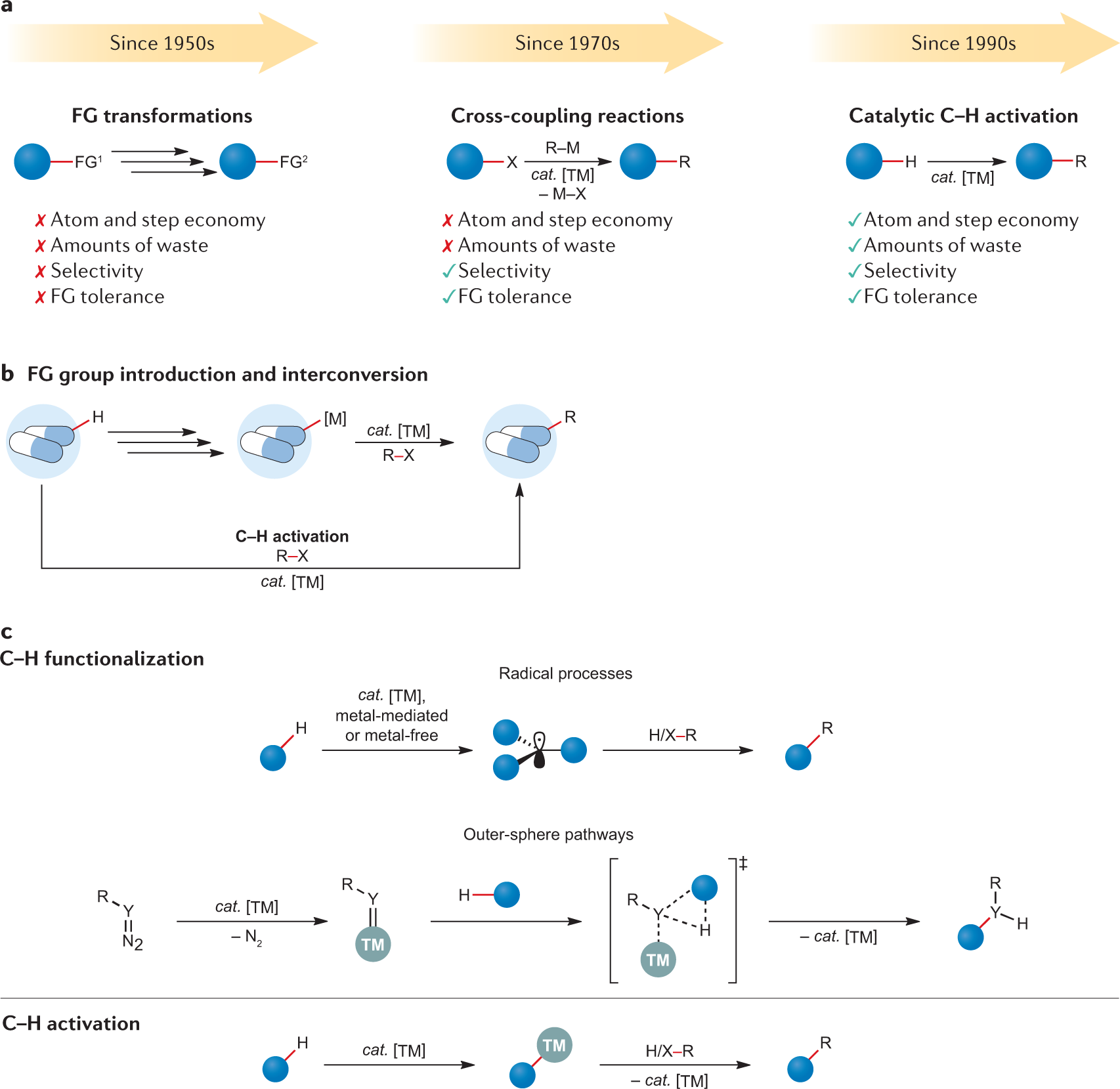
Palladium(II)-catalyzed C-H activation/C-C cross-coupling reactions: versatility and practicality. - Abstract - Europe PMC

Dynamic Kinetic Resolution of Biaryl Lactones via a Chiral Bifunctional Amine Thiourea-Catalyzed Highly Atropo-enantioselective Transesterification. | Semantic Scholar

Kinetics and Mechanism of the Palladium-Catalyzed Oxidative Arylating Carbocyclization of Allenynes | Journal of the American Chemical Society

PDF) Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps
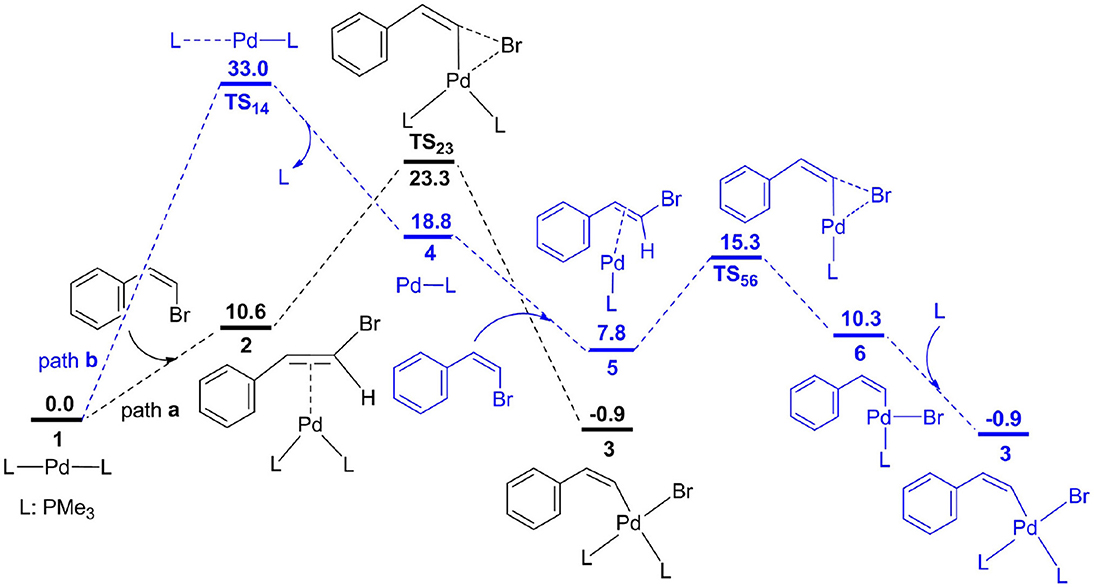
Frontiers | Key Mechanistic Features in Palladium-Catalyzed Methylcyclopropanation of Norbornenes With Vinyl Bromides: Insights From DFT Calculations
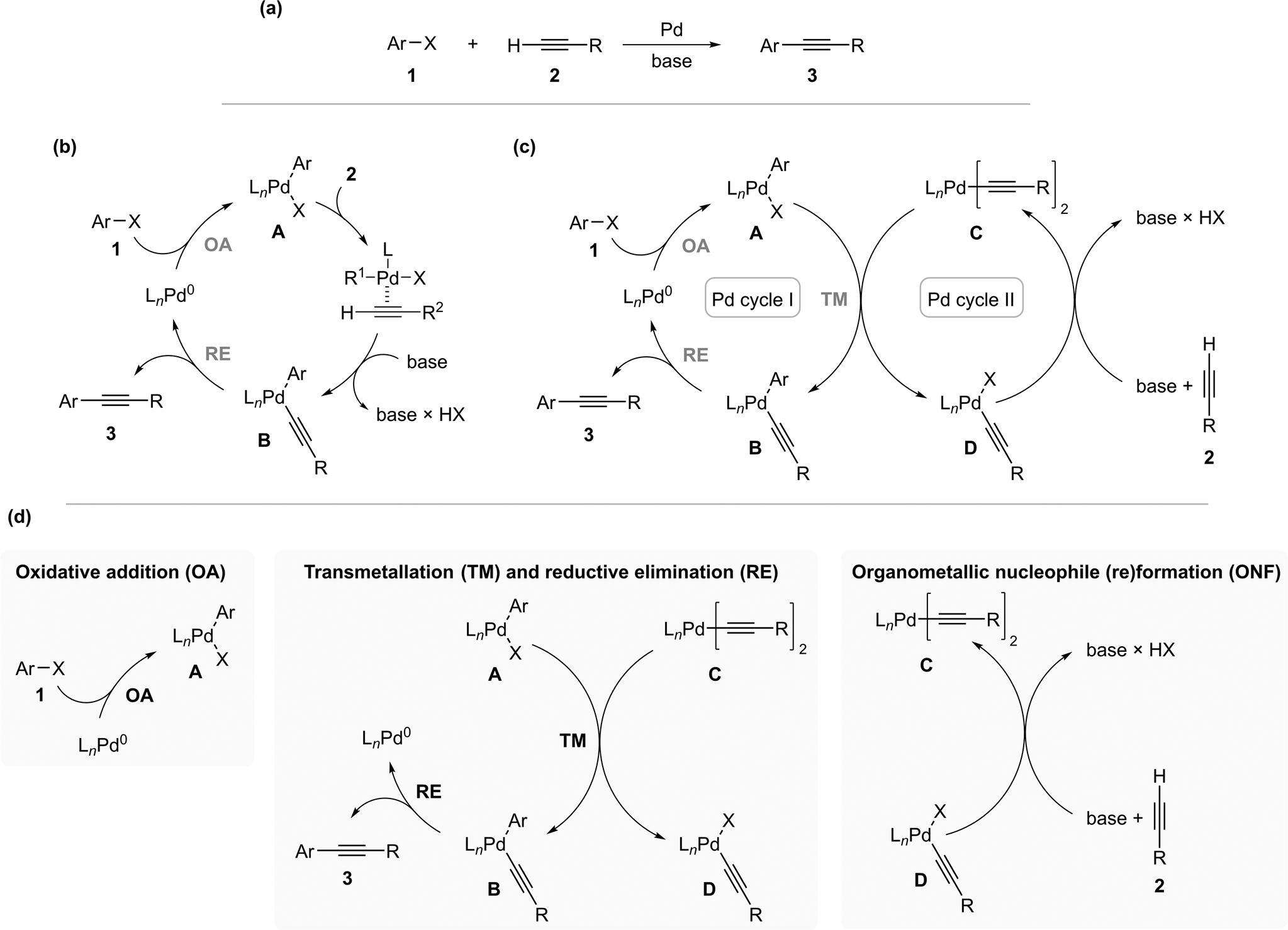
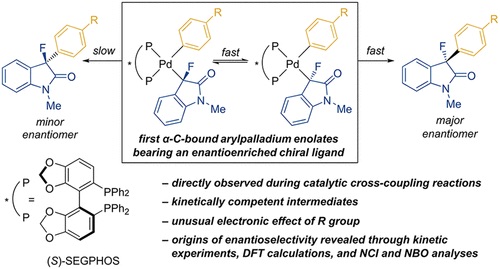
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry
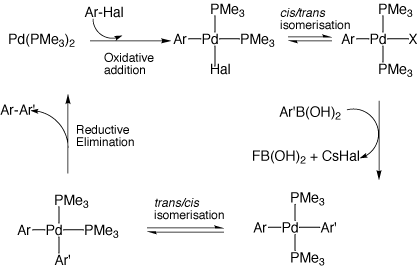
A Computational Study of the Mechanism of Palladium Insertion into Alkynyl and Aryl Carbon-Fluorine bonds

Second Comes First: Switching Elementary Steps in Palladium‐Catalyzed Cross‐Coupling Reactions - Kolter - 2020 - Chemistry – A European Journal - Wiley Online Library

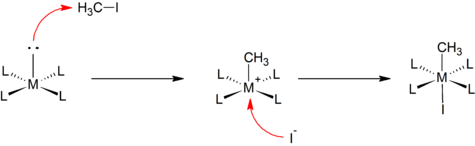
Inorganics | Free Full-Text | Structural Study of Model Rhodium(I) Carbonylation Catalysts Activated by Indole-2-/Indoline-2-Carboxylate Bidentate Ligands and Kinetics of Iodomethane Oxidative Addition

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

Palladium-catalyzed oxidative arene C–H alkenylation reactions involving olefins: Trends in Chemistry
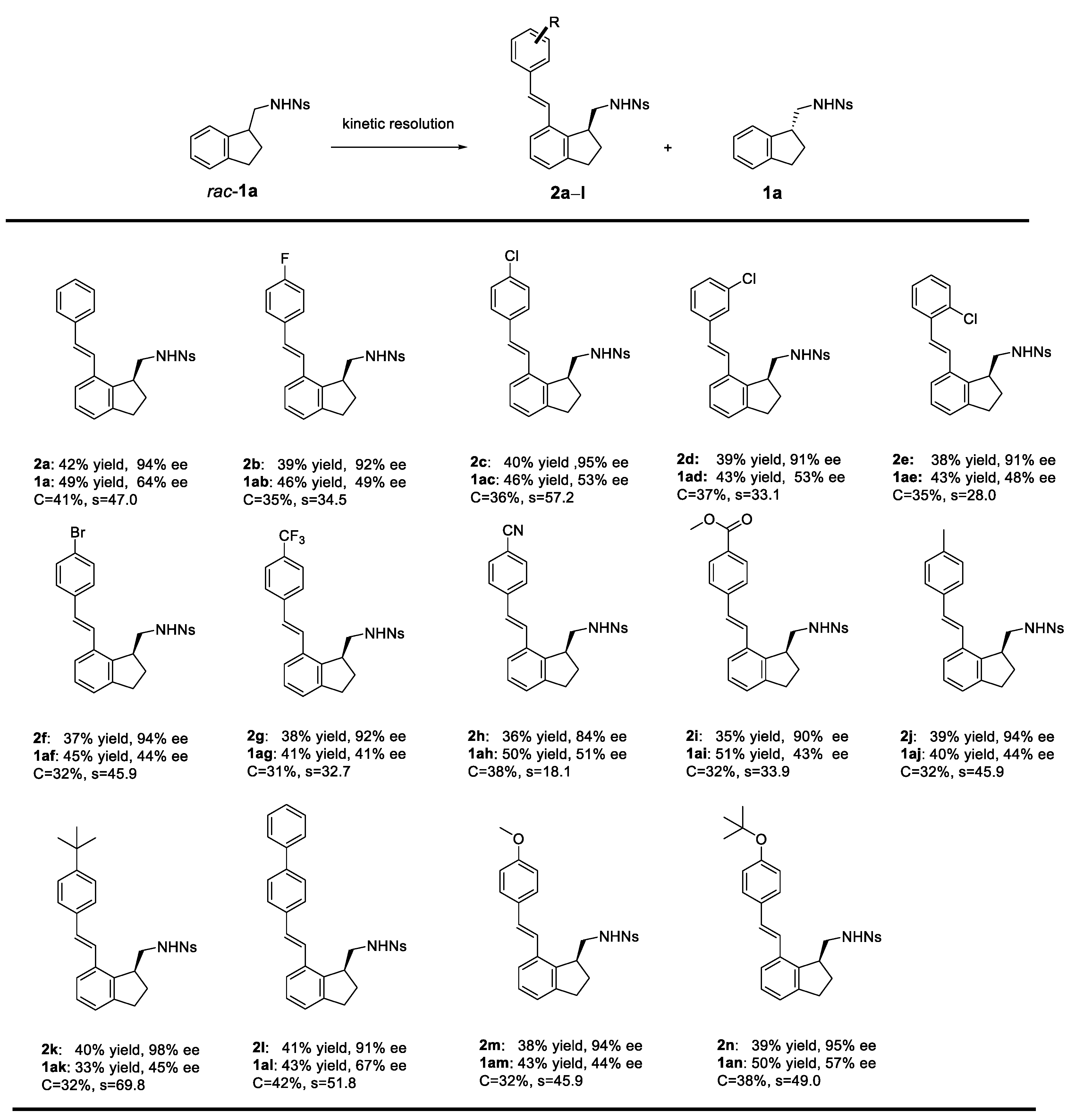
Molecules | Free Full-Text | Kinetic Resolution of β-Alkyl Phenylethylamine Derivatives through Palladium-Catalyzed, Nosylamide-Directed C−H Olefination

A Tutorial on Selectivity Determination in C(sp2)–H Oxidative Addition of Arenes by Transition Metal Complexes - ScienceDirect

Quadruple C-H activation coupled to hydrofunctionalization and C-H silylation/borylation enabled by weakly coordinated palladium catalyst | Nature Communications
Kinetic Studies of the Oxidative Addition and Transmetallation Steps Involved in the Cross-Coupling of Alkynyl Stannanes with Ar